Ratings & Reviews performance provides an overview of what users think of your app. Here are the key metrics to help you identify how your app is rated by users and how successful is your review management strategy.
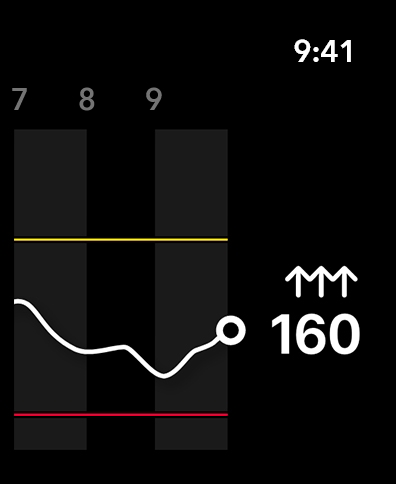
The Simplera™ continuous glucose monitoring (CGM) system helps people manage diabetes. CGM is a technology that lets users view glucose values throughout the day, sends alerts for glucose events on a mobile device, and shows the effects life has on glucose levels. The Simplera™ CGM uses a Simplera™ sensor with Bluetooth™ that converts small amounts of glucose from under the skin into an electronic signal. This signal is used to provide sensor glucose (SG) values to the Simplera™ app on a compatible device. The Simplera™ app provides alerts based on SG levels and the user can enter data such as insulin, meals, exercise, blood glucose (BG) values, and upload information to CareLink™ Personal. The Simplera™ app requires the Simplera™ sensor to function. The Simplera™ CGM provides information that is used for diabetes management but does not provide any direct therapy. In order to access the list of the supported mobile devices and operating systems, go to medtronicdiabetes.com/Device-Compatibility. The clinical configuration of the app is intended for use in clinical research only. It is available only to individuals who have consented to take part in clinical research activities with Medtronic. Clinical studies where this app will be used will follow their own regulatory process and obtain requisite approvals. IMPORTANT NOTE: The Simplera™ system requires a prescription and is indicated for the management of diabetes in persons ages 18 years and older. The sensor is indicated for up to 6 days of use plus an additional 24-hour grace period. The Simplera™ system does not require calibration. Blood glucose (BG) readings are required (1) during the first 12 hours of use, (2) if no sensor data is available, (3) when symptoms do not match the sensor glucose (SG) value, and (4) when taking certain medications. Not taking BG readings as indicated can lead to incorrect sensor glucose readings, over-administration of insulin, and possible hypoglycemia. Proper settings and hearing are also required. Sensor use can also result in skin irritation, bruising, discomfort, redness, bleeding, and infection, and sensors in general also pose a choking risk to young children, which can result in serious injury or death. For complete details see https://www.medtronicdiabetes.com/support/download-library/user-guides and https://bit.ly/SimpleraSafety. This app store should not be used as your first point of contact to resolve technical or customer services issues. In order to protect your privacy and personal information, and promptly resolve any technical or customer services issues you are having with any Medtronic product, please contact your local Medtronic representative. This app is not intended to be a substitute for professional medical advice, diagnosis, or treatment. Always seek the advice of your physician or other qualified healthcare provider with any questions you may have regarding a medical condition or treatment. ©2026 Medtronic. MiniMed and MiniMed logo are trademarks of Medtronic MiniMed, Inc. ™*Third-party brands are trademarks of their respective owners. All other brands are trademarks of a Medtronic company. Apple, App Store, and Apple Watch are trademarks of Apple Inc., registered in the U.S. and other countries and regions. The Bluetooth® word mark and logos are registered trademarks owned by Bluetooth SIG, Inc. and any use of such marks by Medtronic is under license. Other trademarks and trade names are those of their respective owners.